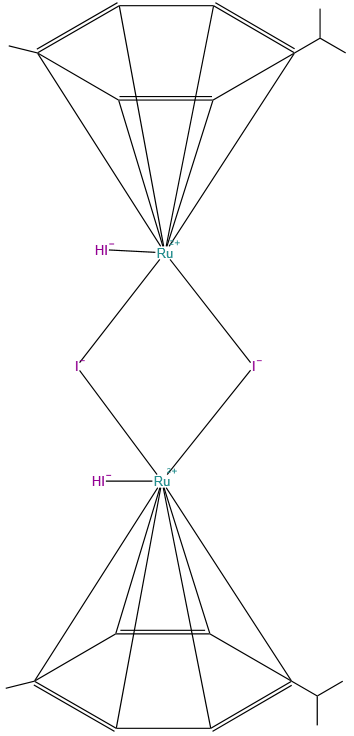
Di-mu-iodobis(p-cymene)iodoruthenium(II)Diiodo(p-cymene)ruthenium(II) dimer
Diiodo(p-cymene)ruthenium(II) dimer
CAS: 90614-07-6
Molecular Formula: C20H22I4Ru2
Di-mu-iodobis(p-cymene)iodoruthenium(II)Diiodo(p-cymene)ruthenium(II) dimer - Names and Identifiers
Di-mu-iodobis(p-cymene)iodoruthenium(II)Diiodo(p-cymene)ruthenium(II) dimer - Physico-chemical Properties
| Molecular Formula | C20H22I4Ru2 |
| Molar Mass | 972.15 |
| Melting Point | 234-236°C |
| Water Solubility | insoluble |
| Appearance | solid |
| Color | red |
| Storage Condition | Keep in dark place,Inert atmosphere,2-8°C |
| Sensitive | Moisture & Light Sensitive |
| MDL | MFCD08064027 |
| Use | A catalyst for many reactions such as ruthenium-catalyzed hydrogenation. |
Di-mu-iodobis(p-cymene)iodoruthenium(II)Diiodo(p-cymene)ruthenium(II) dimer - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R68 - Possible risk of irreversible effects R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36 - Irritating to the eyes R22 - Harmful if swallowed |
| Safety Description | S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S36/37 - Wear suitable protective clothing and gloves. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 2 |
| TSCA | No |
| HS Code | 28439000 |
Di-mu-iodobis(p-cymene)iodoruthenium(II)Diiodo(p-cymene)ruthenium(II) dimer - Reference Information
| Application | diiodo (parvaline) ruthenium (II) dimer is mainly used in asymmetric synthesis reactions, the intermediate [RuI2 p-isopropylbenzene] 2 can be prepared from [RuCI2 p-isopropylbenzene] 2 as a reaction raw material, and further iodinated to produce diiodine (parene) ruthenium (II) dimer. |
| preparation | a) preparation of [RuI2 p-Cumene] 2: 2g of [RuCI2 p-Cumene] 2 and 50 ml of dichloromethane were placed in a flask under nitrogen. 66 mg of tetramethylammonium iodide followed by 10.2 mg of KI in water (50 ml) were added to the solution. The mixture was vigorously stirred at room temperature under inert gas for about 15 hours and the phases were separated. The aqueous phase was extracted with 2 × 40 ml of dichloromethane, the combined organic phases were washed with 3 × 40 ml of water, dried over sodium sulfate, and filtered over carlide. The red-brown solution was obtained and dried under vacuum to give 3.07g of [RuI2 p-isopropylphenyl] 2. B) preparation of diiodo (Paratan hydrocarbon) ruthenium (II) dimer: 155 mg of [RuI2-p-Cumene] 2 and 204 mg of () tmbp was placed in a flask under nitrogen and a mixture of 80 ml of dichloromethane and 30 ml of methanol degassed with nitrogen was added. The mixture was stirred under reflux for 1.5 hours, then it was cooled and concentrated under reduced pressure to give. |
Last Update:2024-04-10 22:29:15
Supplier List
Featured ProductsSpot supply
Product Name: Diiodo(p-cymene)ruthenium(II) dimer Request for quotationCAS: 90614-07-6
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: Diiodo(P-cymene)ruthenium(II) dimer Visit Supplier Webpage Request for quotationCAS: 90614-07-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Diiodo(p-cymene)ruthenium(II) dimer Request for quotation
CAS: 90614-07-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90614-07-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Diiodo(p-cymene)ruthenium dimer Visit Supplier Webpage Request for quotationCAS: 90614-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsSpot supply
Product Name: Diiodo(p-cymene)ruthenium(II) dimer Request for quotationCAS: 90614-07-6
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: Diiodo(P-cymene)ruthenium(II) dimer Visit Supplier Webpage Request for quotationCAS: 90614-07-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Diiodo(p-cymene)ruthenium(II) dimer Request for quotation
CAS: 90614-07-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90614-07-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Diiodo(p-cymene)ruthenium dimer Visit Supplier Webpage Request for quotationCAS: 90614-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



